What does treating a tumor inside the head and searching for subatomic particles have in common? Well, a lot. Targeted cancer irradiation is to be refined by a system of highly sensitive particle cameras. In its design, this futuristic device called Beam TraX resembles the system used by physicists at CERN to understand the internal structure of the fundamental particles of matter. And it uses a camera based on the same technology.
Scientists from the German National Center for Tumor Diseases (NCT), the German Cancer Research Center (DKFZ), and the Heidelberg Ion Beam Therapy Center (HIT) at Heidelberg University Hospital are now testing on the first patients a particle detector supplied by the Czech company ADVACAM. The surprising application of this technology in medicine could help limit the side effects of ion radiotherapy.
“One of the most advanced methods for treating head and neck tumors involves irradiation with ion beams. This has one unique feature: It can be precisely tailored to the depth inside the human head where the particles should have the maximal effect,“ explains Mária Martišíková, InViMo project leader from the German Cancer Research Center (DKFZ)
However, like other types of irradiation, ion radiation also has a catch. The particle beams affect not only the tumor itself but also a tiny part of the healthy tissue around it.
„Such preventive irradiation of the so-called “margin” around the tumor minimizes the probability of the disease’s recurrence. But, it also limits the irradiation of the tumor with a high dose due to the possible side effects such as damage to the optic nerves or memory,“ adds Dr. Martišíková.
Ideally, the irradiation area around the tumor could be narrowed, and the dose to the tumor could be increased. However, current technology does not allow for sufficiently precise targeting of the ions.
This could be addressed by a new device Beam TraX supplied by the Prague company ADVACAM. The „navigation“ of the ion beams inside the head could be improved by tracking some secondary particles created when ions pass through the patient’s head. One could think of this as observing the dust rising behind a fast-moving car.
An insufficiently reliable “map”
A patient undergoing several weeks of ion radiotherapy must first undergo an X-ray computed tomography (CT) scan. The CT image of the inside of the head is essentially used as a “map” to target the tumor with ion beams. According to this “map”, the imaginary “car” drives.
But there is a problem. The situation inside the patient’s head can change during the therapy. The original “map” may differ from the current state inside the skull. This could be due to tissue swelling, tumor shrinkage, or an infection. Until now, physicians lacked a reliable tool to alert them in case of a change in the situation thus reducing the precision of the applied therapy..
Together with ADVACAM, scientists in Germany are now coming up with a promising improvement. Its core consists of particle detectors.
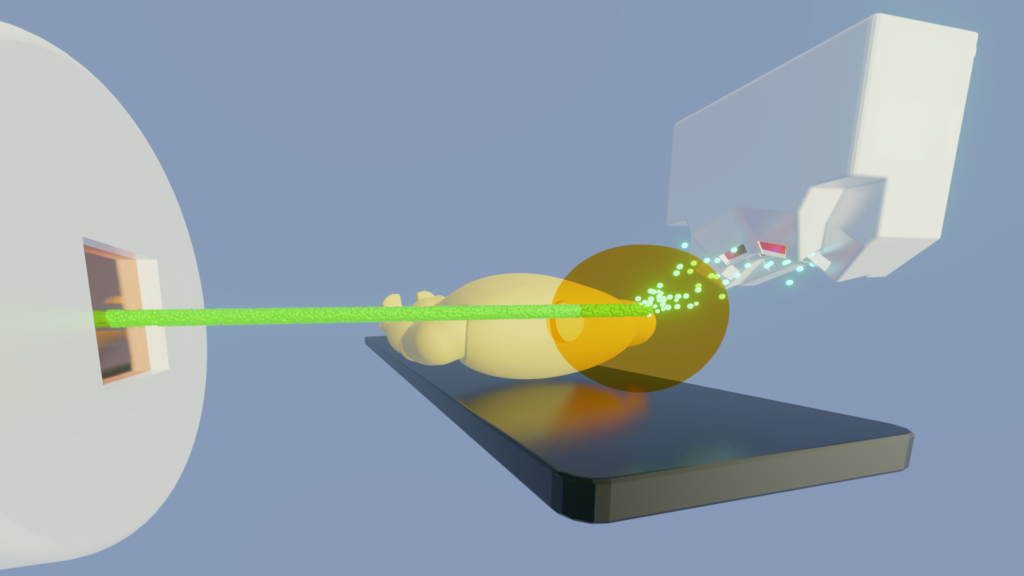
“Our cameras can register every charged particle of secondary radiation emitted from the patient’s body. It’s like watching balls scattered by a billiard shot. If the balls bounce as expected according to the CT image, we can be sure we are targeting correctly. Otherwise, it’s clear that the ‘map’ no longer applies. Then it is necessary to replan the treatment,” describes Lukáš Marek from ADVACAM.
Sparing healthy tissue and enhancing precision
In the fall of 2023, experts in Germany started the clinical study „InViMo“ to verify the potential of this new device. They are focusing on patients with tumors near the base of the skull. This area is challenging to access for irradiation due to the proximity of essential structures like the brainstem.
“We hope the new device will show us how often and where the tumor changes occur. It‘s typically just several millimeters during the therapy. We are also interested in areas where no changes occur or are smaller than assumed. It will allow us to reduce the overall irradiated volume of tissue. At the same time, it will save healthy tissue and reduce the side effects of radiotherapy. We will also be able to apply higher doses of radiation to the tumor. The dose to the healthy tissue will remain below an acceptable limit,” adds Mária Martišíková, the head of the team at the German Cancer Research Center (DKFZ).
The presence of detectors does not affect the existing therapy. It can only benefit from the additional information obtained. In the first phase, data could lead to an interruption and replanning of the irradiation series in case the treatment does not go according to plan. The ultimate goal is a system that would correct the path of the ion beam in real-time.
From particle physics to hospitals
The technology is derived from detectors that helped discover, for example, the famous Higgs boson at CERN. What was initially created for fundamental research is now intended to help treating cancer.
“When we started developing pixel detectors for the LHC we had one target in mind – to detect and image each particle interaction and thereby help physicists to unravel the secrets of Nature at high energies. The Timepix detectors are developed by the multidisciplinary Medipix Collaborations whose aims were to take the same technology to new fields. Many of those fields were completely unforeseen at the beginning and this application is a brilliant example of that,” says Michael Campbell, Spokesperson of the Medipix Collaborations.
Learn More:
Peer-reviewed paper about the metod in Medical Physics journal
Are you curious about how our technology can improve your field?
We are ready to answer any inquiries